Core insights – Cross-sectional systems integration for drug repurposing
Welcome to REPO4EU: Core insights
Our brand new article series where we bring you closer to our project's core innovations and results.
The strategic role of systems integration
A brief introduction
Why does systems integration play such a critical role in REPO4EU and the drug repurposing journey? Quite simply, it is the connecting tissue that ensures every specialized technology and mindset in the project fits together perfectly. By harmonizing diverse elements, we aim to create a comprehensive body of drug repurposing knowledge and workflows that can be seamlessly integrated into a unified Drug Repurposing Platform.
A foundational milestone in this process is the definition of Technology Readiness Levels (TRLs). These benchmarks allow comparisons among the development stages of otherwise disparate technologies. While bridging early scientific discovery with industrial feasibility is usually considered a challenge, establishing workflows to prioritize repurposing candidates can be the key to balance regulatory preparedness and patentability with reimbursement considerations and real‑world unmet medical needs. Ensuring CMC (Chemistry, Manufacturing, and Control) compliance is another essential pillar of our work. Here, specialized templates tailored to the unique regulatory requirements of drug repurposing help streamline the path toward clinical approval.
By connecting these elements, systems integration transforms a collection of standalone tools into a powerful, synchronized engine for drug repurposing acceleration.
The road so far
Scaling technology readiness, AI-powered intelligence and prioritization
At the project’s inception, we established a baseline TRL inventory to map the critical technologies available within the consortium. We have since taken this exercise a step further by projecting the specific TRL requirements needed to meet the platform’s rigorous standards. By extrapolating these interactions, we ensure that every technical component is seamlessly aligned with the platform’s final architecture.
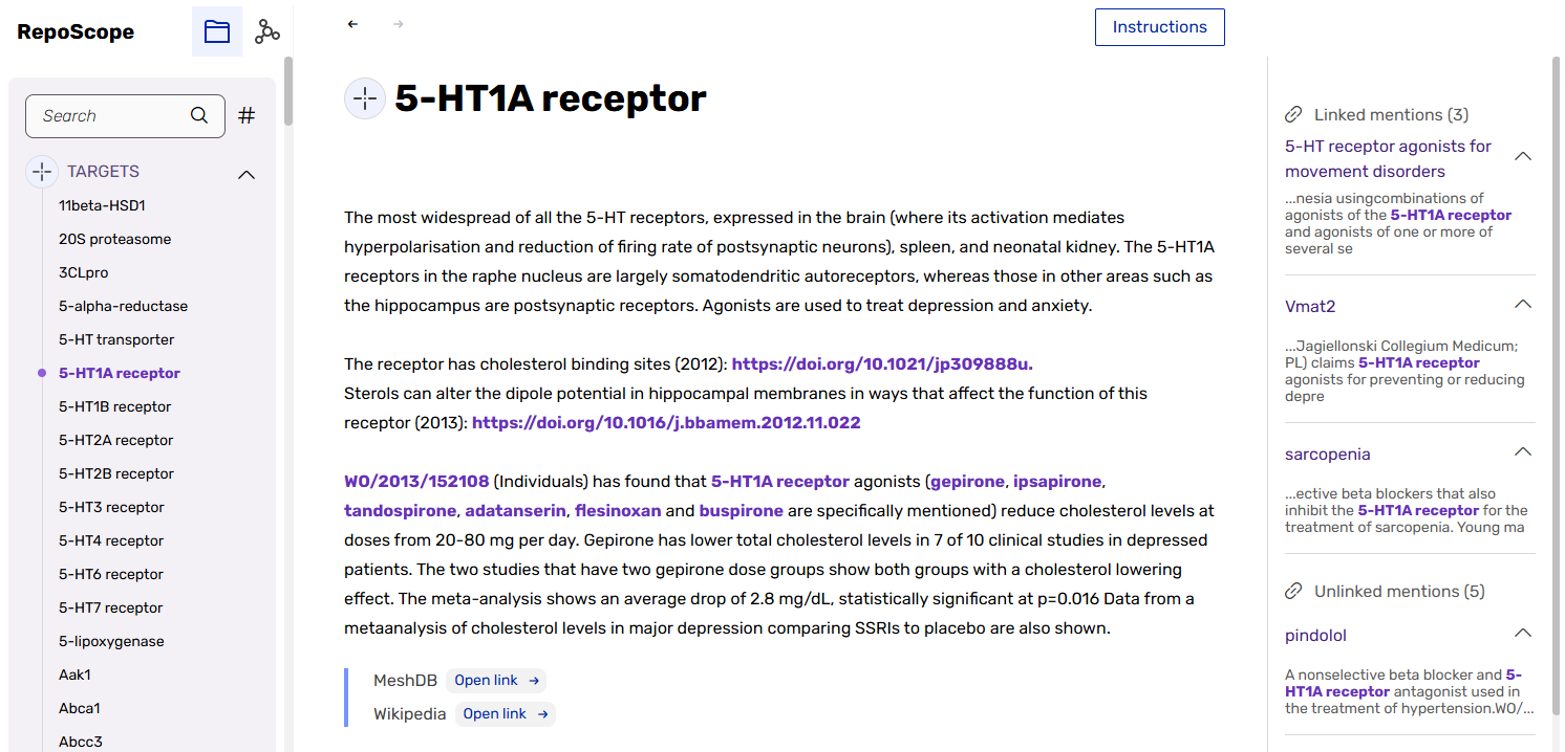
A major milestone has been the provisional integration of the hugely expanded version of the RepoScope knowledge base into the platform’s alpha version, with full integration slated for the beta release.
RepoScope is a database of patents, peer-reviewed papers, conference abstracts, and other scientific information related to drug repurposing. Within the REPO4EU drug repurposing framework, RepoScope serves as a prior art filter for prioritising candidate compounds, enabling users to make educated go/no-go project decisions in a pre-project stage based on what the scientific community has already published, and facilitating freedom-to-operate analyses and strategic patenting considerations.
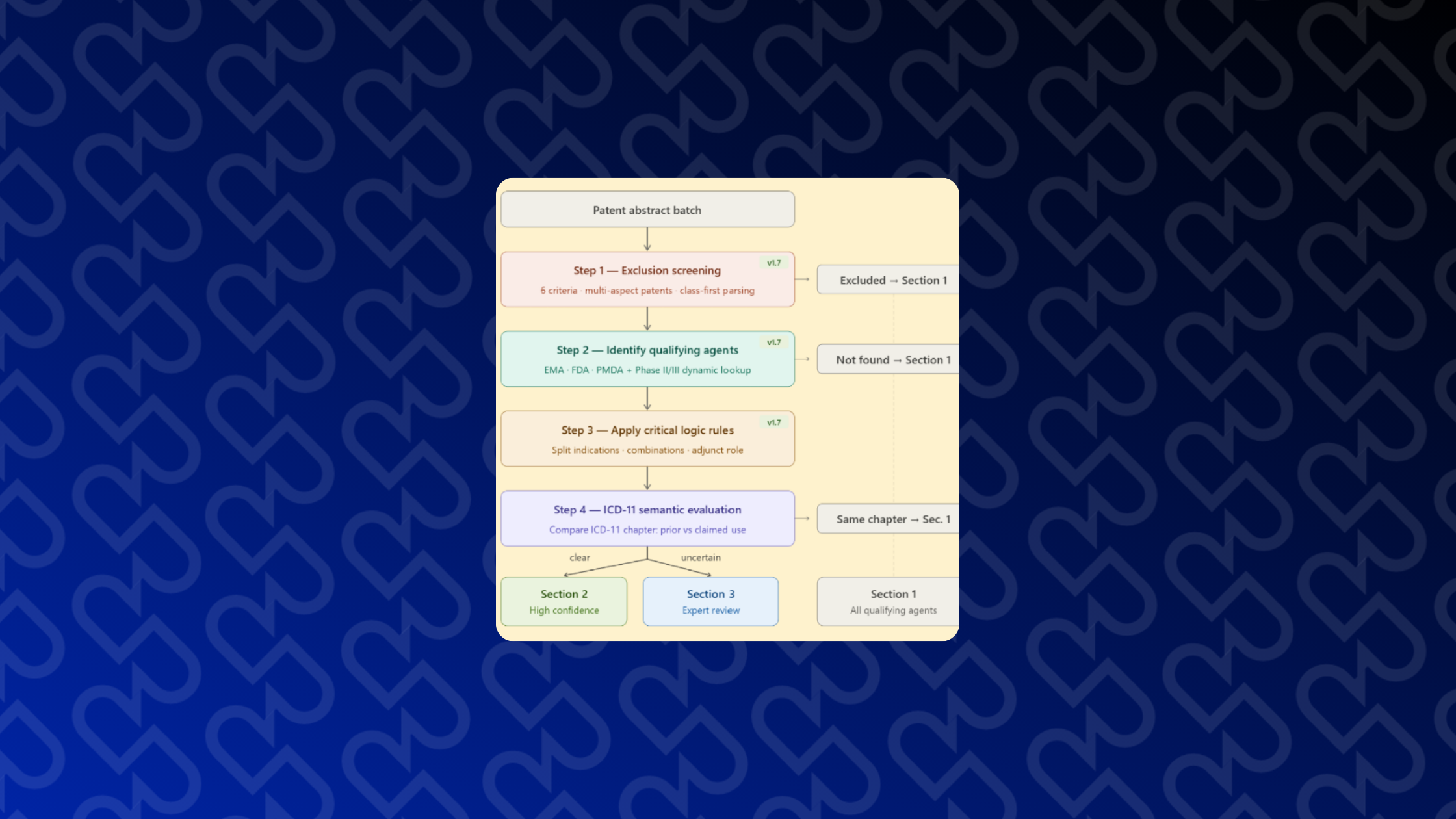
By early 2026, we solved a significant bottleneck: the overwhelming task of manually screening 300–500 life science patent applications per week. This is now handled by RepoDetect, an internal LLM-based screening tool that automates the process with such precision that only a few borderline cases require human review. This advancement ensures the long-term sustainability of RepoScope. Looking ahead, we are training RepoDetect to identify peer-review companion papers, i.e., journal articles already published by the inventors that elaborate on the subject of identified drug repurposing patent applications.
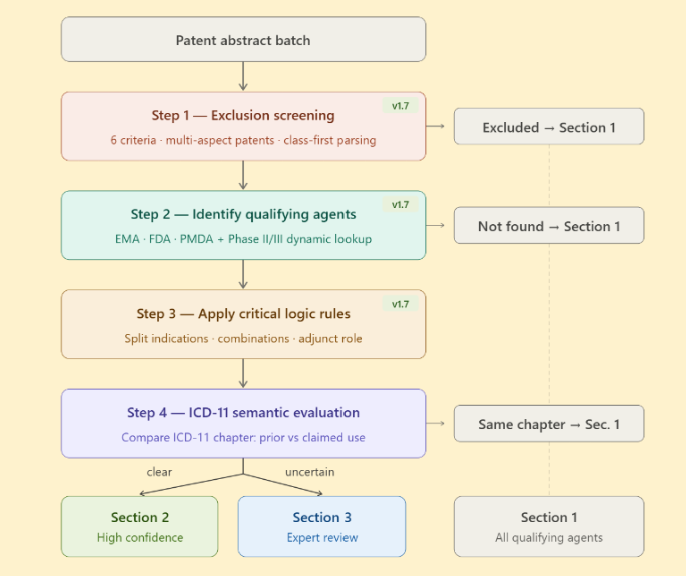
Additionally, meaningful work has been conducted towards building a Unified Prioritization Matrix. One may think of this as a multidimensional decision tree including :
- Multiple “Roots” (Entry Points) defined by the user’s specific repurposing strategy.
- A set of “Common Pathways” for development.
- “Branches” (Exit Points) tailored to the chosen objectives of the drug repurposing project.
To illustrate this in practice, we are developing fictional drug repurposing scenarios that demonstrate how the matrix steers a project from a scientific concept to a viable medical solution.
Looking ahead
Shaping a scalable and responsive infrastructure
Our guiding vision is to architect a scalable, responsive infrastructure that supports EU-wide health readiness and bridges the gap between laboratory discovery and patient care.
As artificial intelligence (AI) continues its rapid evolution, our role is to keep the REPO4EU Platform, the central hub of our drug repurposing infrastructure, at the absolute cutting edge of integrated technology. Because we aren’t just building a static tool; we are cultivating a living ecosystem that adapts to the shifting pulse of modern medicine and technology. Our next milestone is to develop advanced AI support on establishing links to the peer-reviewed paper sphere of drug repurposing.
However, this forward-looking mission is not without its hurdles. While AI has automated much of our screening, the scientifically meaningful abstraction of identified repurposing patent documents and the precise tagging of named entities remains a challenge. Therefore, building a comprehensive knowledge graph of the repurposing patentome requires moving beyond the RepoDetect approach.
As the project’s connective tissue, systems integration demands continuous monitoring of both internal progress and global developments across relevant fields. There is no shortcut for this level of vigilance. It is a fundamental requirement that is as demanding as it is rewarding. Through this ongoing integration effort, we ensure the transformation of fragmented research into a unified, future-proofed engine for drug repurposing.
Behind the scenes
Meet our team of experts
The systems integration pillar is led by HMPC, who is dedicated to bridging the gap between high-level research and clinical readiness. Their work is pivotal in assessing TRLs, developing robust prioritization matrices, and establishing gold-standard CMC documentation guidelines. Beyond technical frameworks, this ensures the project’s resilience by integrating the REPO4EU platform into broader EU health emergency preparedness plans.
With over 25 years of experience spanning both academic research and the corporate pharmaceutical industry, HMPC provides a sophisticated, big-picture perspective. Working closely with the Egnosis and ScienceOpen teams, its leadership ensures an overarching provision of tools and services that harmonizes contributions from our multidisciplinary team of experts, all converging toward a single goal: accelerating drug repurposing.
About our platform
Discover our platform and explore its key features
Become an early tester now!
Follow the link to sign up for our platform's alpha release
Behind the REPO4EU Platform: In conversation with Harald Schmidt (Maastricht University)
Harald Schmidt is a renowned physician, pharmacist and scientific strategist and is one of the leading voices in systems medicine and precision diagnostics. He’s a professor emeritus at Maastricht University, where he acted as a chair of the pharmacology department, and he’s the author of ‘The end of medicine as we know it – and why your health has a future’, where he proposes concrete steps to completely redesign our perception of diseases and how they are treated.
Harald is also a known advocate for mechanism based drug repurposing, a topic he has brought to the forefront of global discussions on medicine and pharmacology as a project coordinator for REPO4EU, the EU-funded initiative that aims to demonstrate Europe’s role as the leader of repurposing efforts amongst the drug innovation arena. In this interview, Harald discusses the ambition behind REPO4EU to build a platform for the global drug repurposing community and how these efforts have the potential to change the future of medicine.
The mission fuelling REPO4EU
Harald, as the coordinator of REPO4EU, can you explain what the core goal of the project is?
Technically, REPO4EU is the response to a funding call from the European Commission to foster drug repurposing across Europe.
Drug repurposing means that instead of developing entirely new drugs, we use medicines that are already on the market and find new uses for them. This has been happening for decades, but usually quite serendipitously; our goal is to make this process much more precise and systematic.
The real innovation of REPO4EU is in how we approach this. We want to fundamentally redefine what we currently call a disease. Most diseases today — except for infectious diseases or some rare diseases caused by a single gene — are defined simply by describing a symptom in an organ. Hypertension, asthma, psoriasis, inflammatory bowel syndrome… these sound like precise disease definitions, but they are really just descriptions of symptoms.
As a result, many therapies are imprecise. Not every patient taking the same medication will experience the same benefit, and this also contributes to the high failure rate in drug discovery.
What we introduce instead are predominantly genetically defined disease definitions, more similar to how rare diseases are approached. This allows us to identify much more precise treatment strategies and opens enormous potential for repurposing existing drugs — not only for their original target, but for completely different disease mechanisms.
Because these drugs are already approved, we already know most of their side effects, and we can skip long discovery and preclinical development stages. That means the process is faster, cheaper, and often much more efficient.
What does coordinating such a large initiative actually involve?
Operationally, my role as a coordinator is making sure this unusually large project — with 27 partners and a €25 million budget — moves in the right direction and that all the different pieces come together successfully.
The real advantage of having such a large consortium is that it allows us to bring together the best people across Europe. What we are doing goes far beyond the initial scientific discovery.
In medicine, we currently face what is often called the “valley of death” — a huge gap between basic research and actual patient benefit. We produce enormous amounts of academic research, but very little of it reaches patients in the form of better diagnostics, therapies or prevention strategies. One major reason is that academic success is still measured mostly by publications and grant income, not by whether your work improves patient outcomes.
REPO4EU tries to bridge that gap. We make sure that from the very beginning, researchers are thinking not only about scientific discovery, but also about patents, regulation, clinical validation, market access and reimbursement. We want researchers to think early about whether their discovery can realistically become something that benefits patients.
That requires bringing together experts from many different areas — science, ethics, regulation, business, intellectual property and healthcare systems. No single person can solve this alone.
Building a one-stop shop for the drug repurposing community
One of the core outputs of the project is the REPO4EU platform. Why is this platform needed, and what makes it so important?
The REPO4EU platform exists because we believe what we are doing will shape the future of medicine.
Rather than waiting for our clinical results to slowly spread through the academic world, we wanted to provide all the tools, guidelines and workflows directly to researchers so they can apply these methods themselves.
One of our biggest achievements is in bioinformatics. Traditionally, to work with bioinformatics, you needed to be a bioinformatician — someone who could code and use highly specialised software. We wanted to create something that the average biomedical scientist could use without needing that expertise. That required a lot of work because bioinformaticians and biomedical scientists often speak almost different languages.
We now have a very strong tool called drugst.one that makes this possible. Instead of generating a long list of 100 possible drug candidates, we aim for extreme precision — ideally narrowing it down to just two or three highly relevant options.
Beyond that, the platform integrates everything needed for the full translational process: preclinical validation, access to human cell models, clinical trial design, patent strategy, regulatory guidance, reimbursement planning and health technology assessment. It’s not just a collection of links or tools — it’s a fully integrated online workspace that guides researchers through the entire journey from idea to patient impact. And importantly, it can be used globally, not just in Europe.
Why is an interdisciplinary approach needed to develop this platform?
Nobody in this project works in isolation. You cannot simply sit in your office, do some bioinformatics, and hand the results over to someone else. The process is highly iterative and requires constant dialogue between teams.
For example, after bioinformatics predictions are made, we immediately need discussions around preclinical validation. Can we validate using human cell models? Do we need animal models? Is it safe enough to move directly into clinical trials?
At the same time, we apply an ethics by design approach. We do not want ethics to be something that is considered at the very end, just before a clinical trial starts. Ethics should be part of the process from day one. The very first question should be: is there really an unmet patient need? Does this approach genuinely benefit patients? Are the risks acceptable? If the answer is no, then there is no reason to continue with further experiments.
Likewise, patent strategy and commercial viability must be considered early. If something cannot realistically be developed into a therapy, then even excellent scientific results may end up as a translational dead end.
Everything we do is built around one central goal: not papers, but patients.
Looking ahead — enhancing the platform’s capabilities
The beta version of the platform will be released in the autumn of 2026. What do you expect from this milestone?
Two things. First, that the platform truly works — that it can reliably guide discoveries toward real patient impact; and second, that it is widely used by scientists, clinicians, biotech companies and industry partners.
The goal is scale. We want to accelerate innovation in diagnosis, treatment and prevention as quickly as possible.
Drug repurposing is only one part of this. Precision prevention is another major area. If we can better define disease risk, we can also provide much more personalised prevention strategies, reducing the need for treatment later on. In many cases, lifestyle interventions may be enough if they are tailored precisely enough. Only in a smaller percentage of cases would drugs be needed. So this is really a broader shift toward precision medicine as a whole.
Right now, we are preparing both alpha and beta testing phases of the platform, and feedback is incredibly important. We invite researchers, clinicians and other stakeholders to register via this link, so they can test the platform and tell us what works and what needs improvement.
Finally, who do you envision will be using the platform once it is fully launched?
A very broad range of stakeholders!
Bioinformaticians may want to better understand what happens after the computational predictions. Biomedical scientists can use the tools directly. Biotech companies, pharmaceutical companies, regulators, health technology assessors — they all have a role to play.
Even patient organisations are very important for us. While we do not make specific recommendations for individual patients, we absolutely want patient groups to engage with us and help shape the process.
I think everyone involved in healthcare innovation can benefit from looking a little outside their own silo and understanding how the whole system works. That is exactly what REPO4EU is trying to build.
REPO4EU: The Podcast
Our podcast brings listeners closer to the latest innovations, research and developments happening in drug repurposing across the globe. In the second season, ‘Platform Conversations’, we're exploring the process of building an online platform that will bring everything to do with drug repurposing under one roof — made in Europe with global reach. We will hear form the experts involved in making this ambition a reality.
Core insights – Pre-clinical validation of scientific questions
Welcome to REPO4EU: Core insights
Our brand new article series where we bring you closer to our project's core innovations and results.
Preclinical validation
A brief introduction
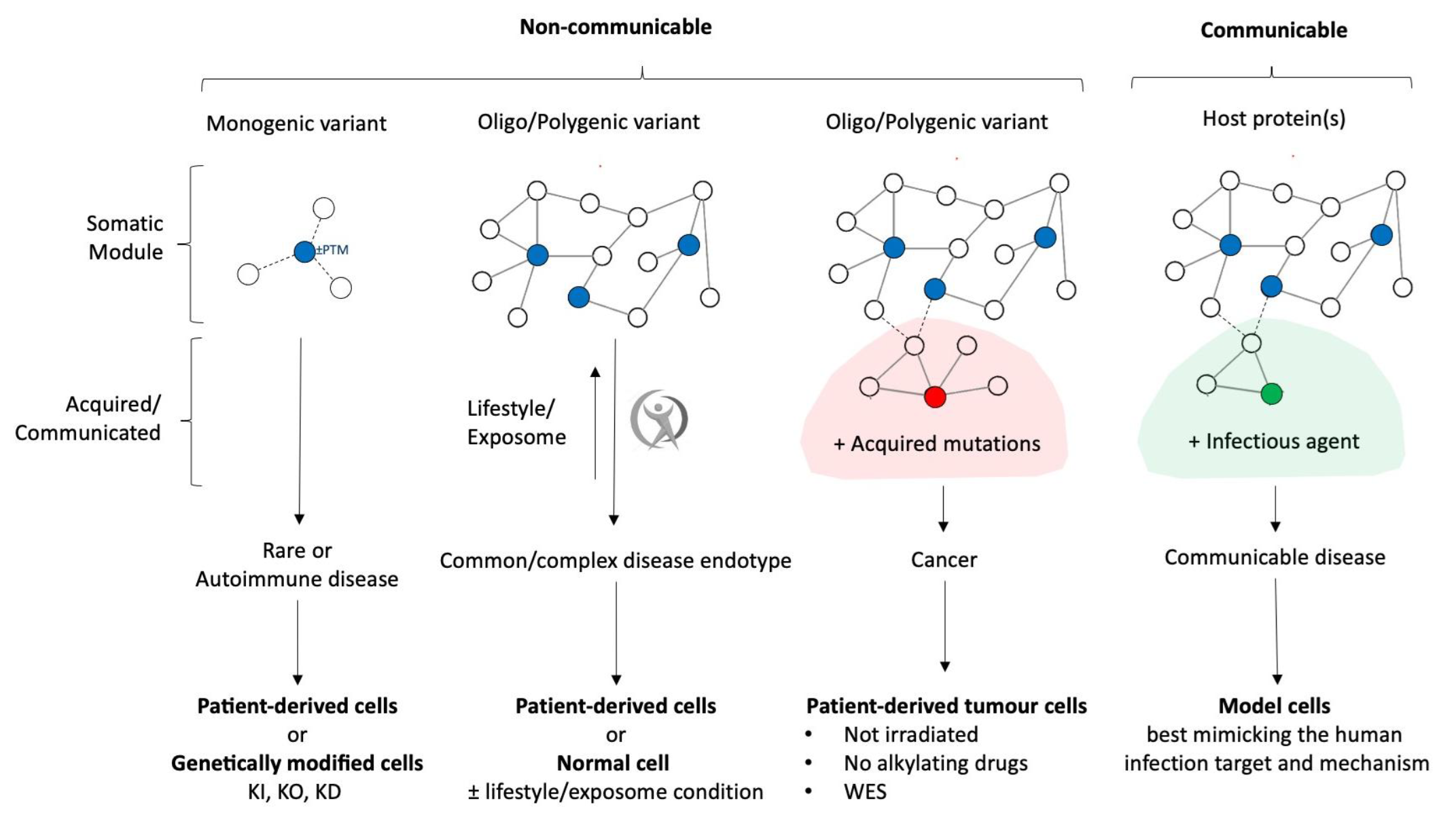
In the pharmaceutical world, clinical trials play the role of the ultimate “reality check”. However, they are incredibly expensive, time-consuming and involve the most precious variable of all: human lives. Thus, de-risking the path to clinical phase via effective pre-clinical validation lies at the heart of REPO4EU’s ambition.
Within the REPO4EU project, we aim to secure a massive head start for all repurposing projects by providing robust validation of the preclinical hypotheses, generated through advanced bioinformatics, in silico-based target identification, and computational drug predictions. Preclinical validation moves a prediction into living systems, and this exact bridge between computational approaches and mechanistically relevant cellular and animal models, which mimic human disease endotypes, ensures a strong scientific foundation for each drug candidate.
Our goal is to identify the most promising drug repurposing opportunities and ensure that only rigorously validated candidates advance to clinical stages. This approach builds a more precise, safer, and faster highway, accelerating the path from computational discovery to real-world therapeutic impact, while also prioritizing patient safety.
The road so far
From digital blueprints to validated biological evidence
REPO4EU envisions a future where drug repurposing evolves into a precise, mechanism-driven discipline, firmly based on validated biological evidence. The progress achieved so far through database development, validation pilots, and methodological guidelines, directly supports this vision.
One of our most significant milestones to date is the development of a comprehensive open-access, user-friendly database of cellular and animal disease models, specifically designed to capture the ones with the highest translational potential. This resource acts as a “compass” for researchers, helping them to pinpoint the most appropriate models for validating computational predictions. By strengthening the bridge between in silico predictions and experimental testing, we ensure that the transition is both seamless and scientifically sound.
To demonstrate the power of our approach, REPO4EU has launched validation pilots in three high-stakes medical areas: thyroid cancer, diffuse pontine glioma (DIPG), and infectious diseases. These pilots serve as an essential testing phase providing clinically relevant proof-of-concept studies on repurposing hypotheses before moving to the clinical trials.
Beyond data and testing, we have also developed methodological guidelines for identifying the most appropriate disease models (both cellular and animal), selecting promising repurposable drug candidates, and defining the rationale for combination therapy strategies using a network‑pharmacology approach. Fostering a culture of collaboration, transparency and FAIR (Findable, Accessible, Interoperable and Reusable) data that accelerates the translation of preclinical findings into life-saving clinical scenarios, the guidelines will be made openly accessible to the global scientific community.
Looking ahead
Towards a robust preclinical validation framework
REPO4EU’s commitment to a transformed medical landscape remains the primary force guiding our work. By systematically connecting bioinformatic predictions with clinically relevant models, we are laying the groundwork for an open-access and scientifically rigorous pipeline. Within our platform, the flagship output of our project, stakeholders will be able to access validated tools, disease models, and decision‑making frameworks that bridge the critical gap between computational discovery and clinical application.
Through this infrastructure, we ensure that repurposing strategies are not only innovative but also precise, reproducible, and ready for regulatory consideration. We are redefining the standards for preclinical validation across Europe and beyond, moving away from fragmented research toward a unified, transparent ecosystem.
And we remain laser–focused on our long-term vision, where drug repurposing accelerates therapeutic development, reduces costs, mitigates risks, and ultimately delivers patient-centered solutions to some of the most pressing health challenges worldwide.
Building on the significant progress achieved so far, we embrace the work required to maximize translational impact as an opportunity for further growth and innovation. Maintaining the translational relevance of disease models and ensuring that cell and animal systems reflect the complexity and diversity of human disease, remains one of our central scientific imperatives. Equally important is the integration of heterogeneous data sources, from computational predictions and preclinical models to clinical datasets, which calls for advanced frameworks for harmonization and interpretation. Another crucial step toward enabling downstream clinical validation is effectively aligning methodologies with regulatory requirements.
Beyond these structural challenges, the scientific landscape itself is constantly evolving. Advances in disease endotyping and in our understanding of drug mechanisms demand ongoing methodological refinement to ensure that preclinical strategies remain up to date and clinically meaningful. Addressing these challenges will be essential to ensure we continue delivering robust, safe, and translatable outcomes that advance the goals of REPO4EU.
Behind the scenes
Meet our multidisciplinary experts’ alliance
The strength of REPO4EU lies in its collaborative engine bringing together diverse expertise to transform computational theories into clinical realities.
Our preclinical validation team, led by Maastricht University, leverages their leading expertise and long-standing experience in preclinical experimentation and animal models to drive reliable validation of drug repurposing candidates. RUMC and UZH contribute specialized knowledge in thyroid cancer and DIPG, providing both animal models and clinical insights that support scientific translation. UHAM contributes essential expertise in viral infectious disease models, while BioLizard ensures methodological statistical rigor to support reliable results. To bridge preclinical data with clinical application, HMPC and 3DPX play a key role in supporting the clinical translation of findings. Finally, GeneSurge is responsible for integrating complex datasets and guiding scientific decision-making.
Collectively, we form a multidisciplinary framework that ensures evidence-based progression of drug candidates.
About our platform
Discover our platform and explore its key features
Become an early tester now!
Follow the link to sign up for our platform's alpha release
Core insights - Transforming drug repurposing through real-world data-driven Artificial Intelligence
Welcome to REPO4EU: Core insights
Our brand new article series where we bring you closer to our project's core innovations and results.
What is real-world data-driven Artificial Intelligence?
A brief introduction
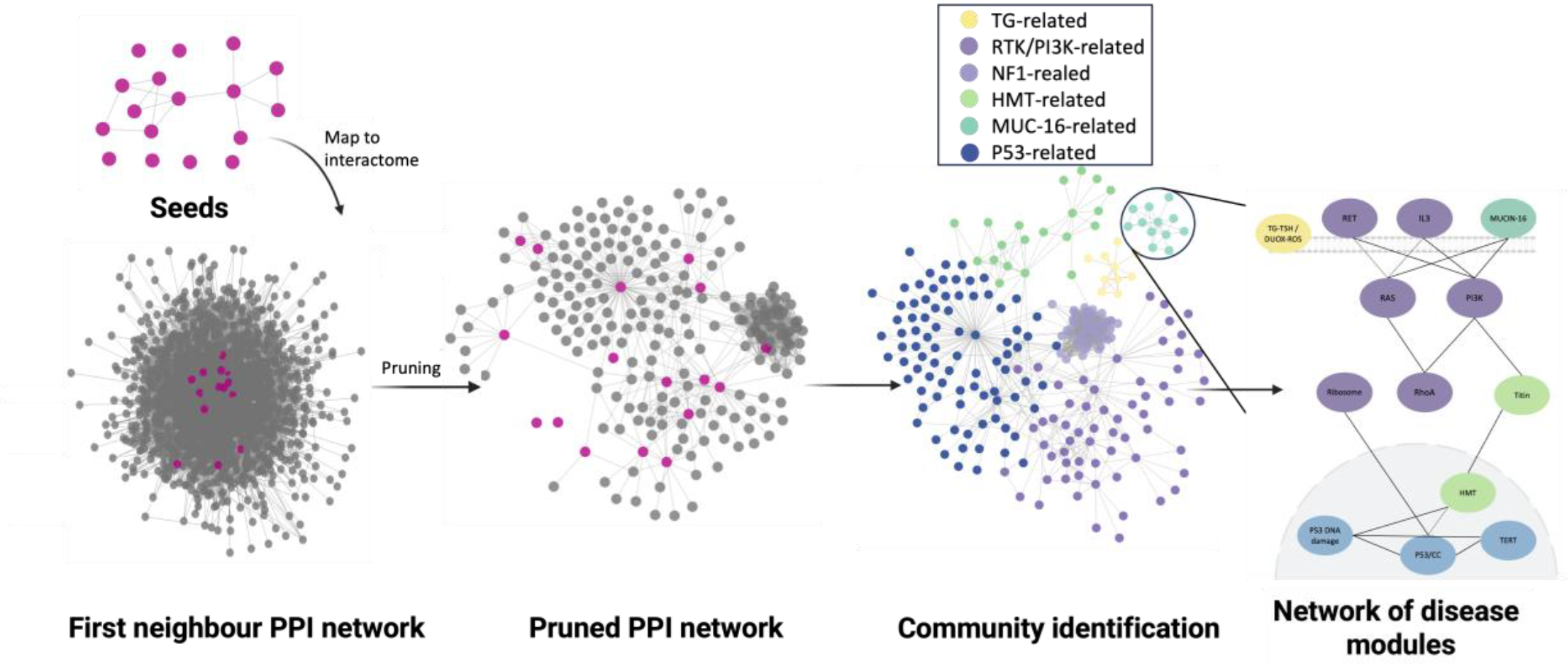
Artificial Intelligence (AI) has undeniably emerged as the definitive revolution of our era, fundamentally reshaping the global landscape by collapsing the distance between human intent and execution. Within the REPO4EU project, AI is far more than a buzzword; it represents a profound paradigm shift. By synergizing AI with systems biology and network pharmacology, we aim to move beyond symptom-based treatments to redefine diseases through mechanism-based endotypes, generating robust, data-driven hypotheses for drug repurposing.
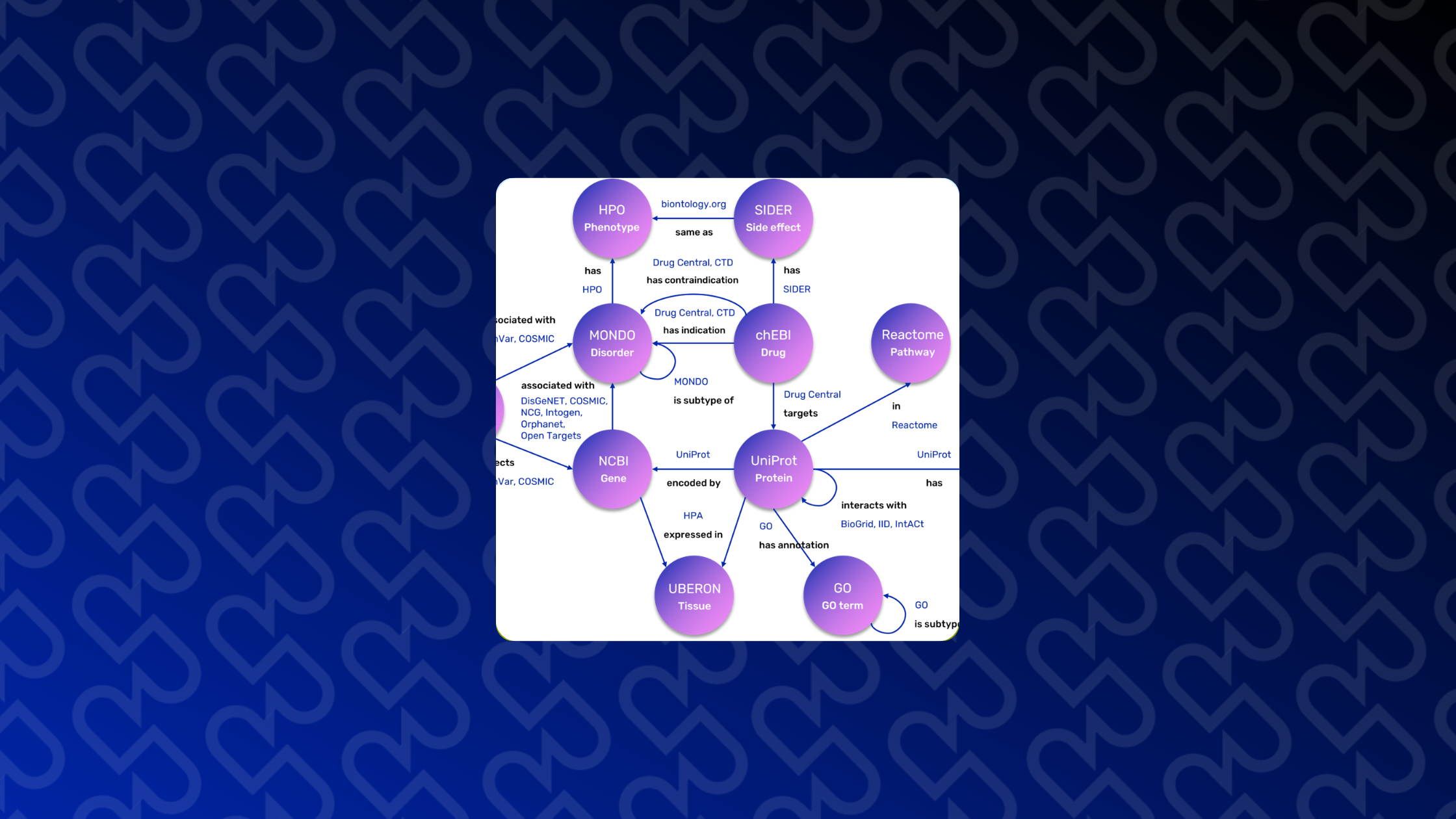
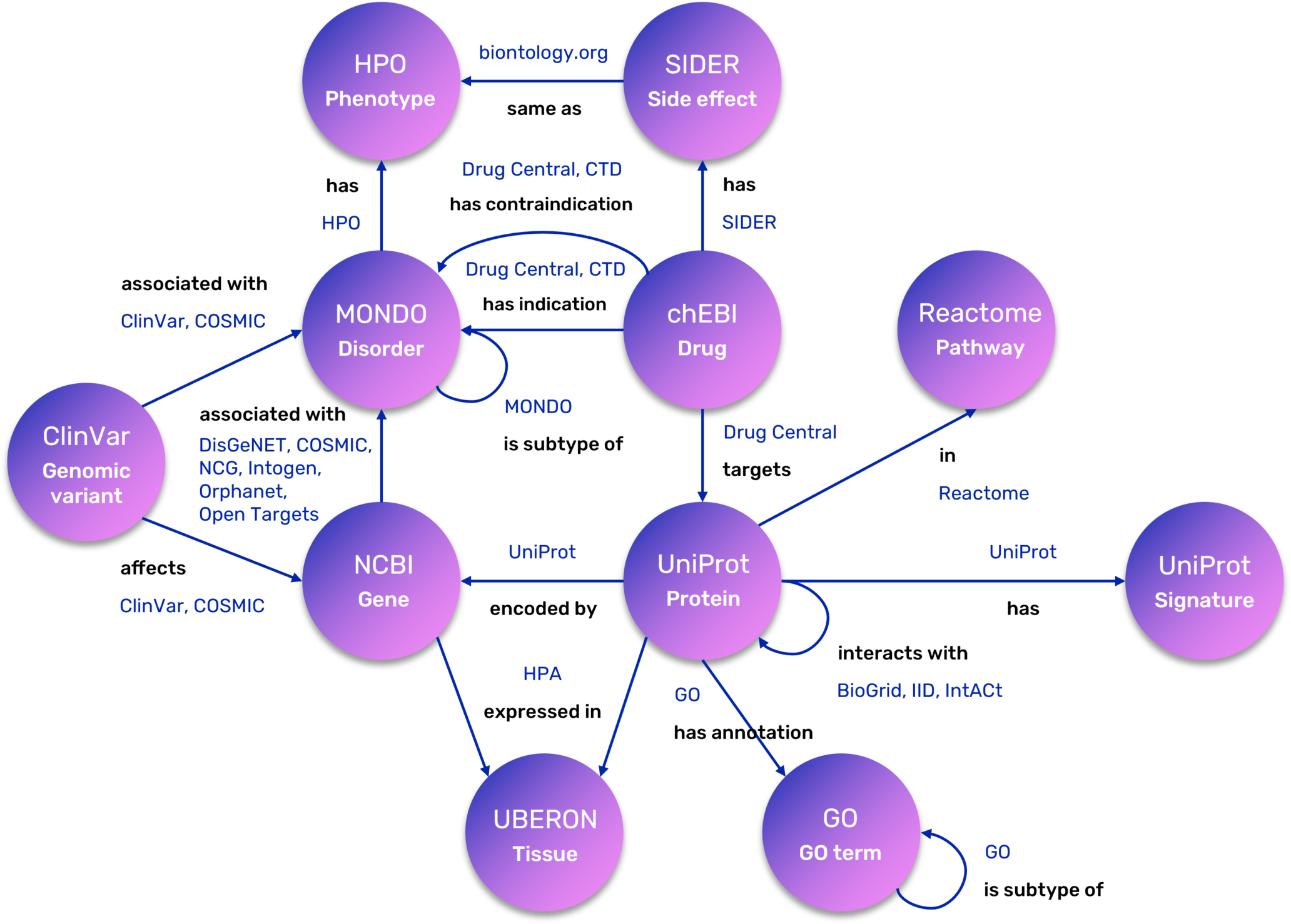
The complexity of this field requires the seamless integration of vast, heterogeneous data: from drug targets’ information, signaling pathways and protein-protein interactions to gene-disease associations, gene expression, and adverse drug effects. Consequently, REPO4EU is developing a comprehensive knowledge base aiming to bring these diverse data sources together into a harmonized, interoperable framework that supports downstream analyses and decision-making. This framework serves as the foundation for uncovering the molecular mechanisms of diseases and identifying promising drug targets and compounds for repurposing. These hypotheses are further refined through advanced in- silico workflows and subsequently validated in pre-clinical and clinical studies.
The road so far
Towards a robust repurposing framework powered by AI
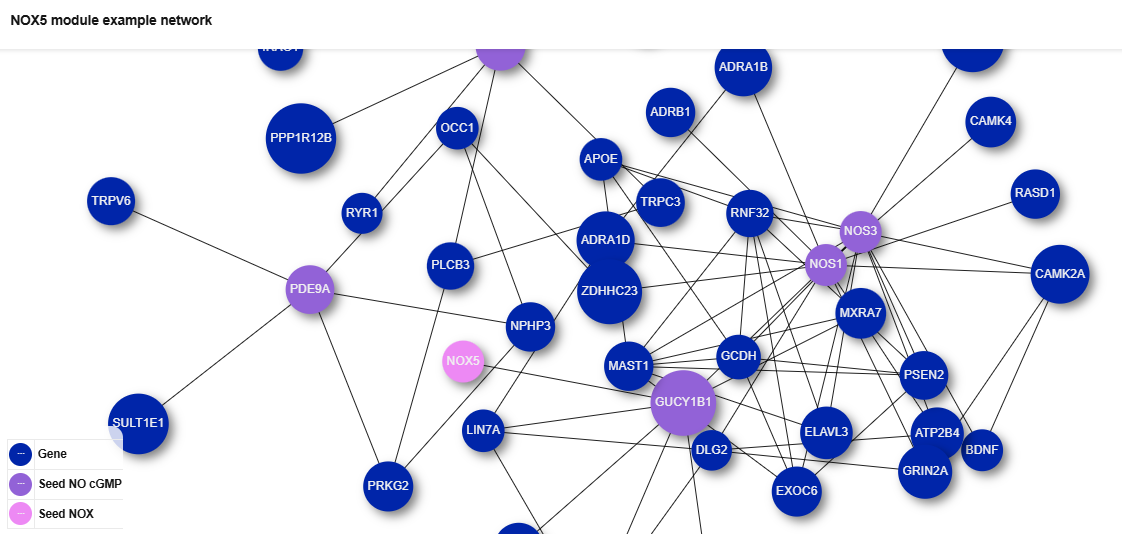
A key achievement of the REPO4EU project is the expansion of the NeDRex database, which serves as our foundational knowledge base and is an integral part of the REPO4EU platform. This extensive, continuously updated resource provides the structural backbone for downstream analyses. To ensure our framework remains at the cutting edge of computational drug repurposing, we carried out an extensive literature collection, review, and categorization, now maintained and expanded through automated search queries and specialized AI-driven tools.
Building upon this data-rich foundation, we have developed a sophisticated suite of computational workflows and software tools, which includes:
- A fully automated pipeline for disease module inference and drug repurposing hypothesis generation.
- A dedicated chemoinformatics pipeline.
- A tool for selecting high-confidence, disease-associated gene sets
- A drug repurposing tool leveraging Graph Neural Networks (GNNs), as well as a web-based application for refining and exploring putative disease mechanisms.
Collectively, these advancements empower a continuous cycle of discovery, enabling the generation of high-precision drug repurposing hypotheses that are systematically refined and validated through a rigorous integration of computational and experimental evidence.
Looking ahead
Envisioning new industry standards
As we move forward, REPO4EU remains committed to a vision where AI and bioinformatics are not merely experimental additions but foundational pillars of the drug development pipeline. Our ultimate goal is to embed these technologies into the industry standard, enabling the identification and validation of mechanism-based, patient-tailored therapies with unprecedented speed and accuracy.
However, the path to a standardized model is defined by its challenges as much as its opportunities. Mechanistic drug repurposing is inherently complex, requiring a sophisticated interdisciplinary blend of expertise, advanced computational methods, and access to high-quality, diverse datasets. To generate meaningful in-silico hypotheses, we must maintain access to high-quality, diverse, and harmonized datasets —a task made more difficult by the fact that disease mechanisms and patient responses are highly idiosyncratic. Furthermore, the rapid evolution of the AI field demands constant iteration and requires constant adaptation of our tools and methods. Critically, while AI acts as a powerful catalyst for discovery, it should not become a replacement for human expertise, but a collaborative tool that augments the researcher’s ability to navigate the biological maze instead.
To overcome the challenges and stay true to our vision, we must and will continue refining our tools and workflows and foster collaboration across disciplines, to ensure that precision drug repurposing evolves from a specialized research effort into a routine, life-saving component of global clinical innovation.
Behind the scenes
Meet our Experts’ team
A multidisciplinary powerhouse of academic, clinical, and industry partners (STALICLA, University of Hamburg, University of Vienna, Brigham and Women’s Hospital, Technical University of Madrid, BioLizard, Maastricht University, Radboud University Medical Center, University of Zurich), led by the Technical University of Munich, contributes specialized expertise to the development of the REPO4EU AI-driven repurposing framework.
By integrating foundational data with specialized expertise in patient stratification and chemoinformatics, infectious diseases, and rare disease mechanisms, we transform fragmented information into actionable medical intelligence. This synergy is further sharpened by advanced in-silico validation and clinical endotyping, ensuring every hypothesis is grounded in real-world plausibility.
Together, we form a cohesive ecosystem dedicated to bridge computational modeling, clinical insight, and translational application towards a new life-saving era for patients.
About our platform
Discover our platform and explore its key features
Become an early tester now!
Follow the link to sign up for our platform's alpha release
RExPO25: The 4th International Conference on Systems Medicine, AI & Drug Repurposing
On 24-26 September 2025, representatives from the entire drug repurposing ecosystem gathered in Barcelona for RExPO25, the fourth edition of the international series of conferences organised and hosted by REPO4EU.
Under the premise of being at the heart of latent discussions shaping the future of medicine, this year’s conference brought together leading voices from academia, industry, policy, HTA bodies and patient-focused organisations to explore how systems medicine, artificial intelligence and innovative regulatory and reimbursement approaches can accelerate drug repurposing for the benefit of patients across Europe.
Building on the momentum of previous editions, RExPO offered once again a truly interdisciplinary programme, reflecting on how far the field has matured and how close repurposing is to delivering real-world clinical and societal impact, according to RExPO25 Co-chairs Harald Schmidt, Head of Pharmacology at Maastricht University and REPO4EU Coordinator, and Emrey Guney, CTO and Head of Discovery & Data Science at STALICLA.
“Over the years organising this conference, we have learned a lot about the challenges, the use cases and the stakeholders involved. We are now entering a phase where we see validations, specifically in cancer and Alzheimer’s trials, and the focus is increasingly on precision and clinical proof. This year, we really tried to cover all of this in a broad and interdisciplinary manner,” explained Prof. Schmidt.
From computational discovery to clinical validation
The RExPO25 agenda spanned the full drug repurposing pipeline, from advanced computational methodologies and systems biology approaches, to clinical validation and implementation.
Over 60 international speakers, spread across 15 sessions, delivered talks highlighting how AI-driven modelling, multi-omics integration and network-based approaches are enabling researchers to identify new therapeutic indications for diseases that have yet to find a viable cure – one that targets the root cause, and not merely treats its symptoms.
Several speakers emphasised that the field is now moving beyond proof-of-concept towards tangible clinical evidence, but also stressed the importance of maintaining robust, trustworthy frameworks when applying AI in biomedicine.
“We are living in an incredible moment where many developments are crystallising into reality. The computational capacity in Europe is growing, and artificial intelligence is at the centre of this, but we must keep an equilibrium — with trusted frameworks, scientific rigour and an understanding of real clinical constraints,” said Alfonso Valencia, Director of the Life Sciences department at Barcelona Supercomputing Center.
“We need drugs to treat diseases, but developing drugs is a complex challenge. This is where the idea of repositioning drugs from one indication to another is extremely powerful, but if we want to do this in a more effective way, we still need more knowledge,” added Prof. Valencia during his RExPO25 Honorary Lecture.
Regulation, reimbursement and access: confronting the real hurdles
Beyond scientific and technological advances, RExPO25 placed strong emphasis on the role of regulation, health technology assessment (HTA), reimbursement and market access — which are deemed amongst the community as some of the biggest hurdles for bringing repurposed medicines to patients.
Some of the experts representing payers and HTA bodies brought critical perspectives to the conversation. For instance, Sibylle Reichert, Executive Director at International Association of Mutuals (AIM), reinforced the fact that protecting universal access to healthcare is crucial in Europe, which means medicines must remain affordable, accessible and available. “That’s why reimbursement organisations are such important players in this space,” she said.
Following this thread, the discussions underscored that science, regulation and reimbursement cannot be treated as separate worlds, as Anja Schiel, Senior Advisor at Norwegian Medicinal Products Agency (NOMA), put it: “You cannot disconnect science from reimbursement decisions. Repurposing is a no‑brainer when it comes to savings and equity, but collaboration and knowledge transfer across stakeholders is essential.”
Also reflecting on the need to balance accessibility with incentives for innovation, Lynn Durham, CEO and Founder of STALICLA, highlighted that researchers operating in this space believe in the common good, but they operate in a system that is strongly tied to regulation and private investment.
“That’s why we need to strike a balance between accessible pricing and acceptable returns, so that drug repurposing can be sustainable,” Durham added.
Looking ahead — together
A recurring theme throughout RExPO25 was the need for systems-level thinking and collective innovation. Speakers repeatedly highlighted that no single discipline, sector or stakeholder can advance drug repurposing alone. This collaborative spirit was reflected not only in the programme but also in the openness of discussions across collectives that have traditionally worked in silos.
As the fourth edition of RExPO concluded, one message was clear: drug repurposing is no longer a niche concept, but a powerful and increasingly validated strategy to address unmet medical needs more efficiently. The challenge now lies in scaling these successes through coordinated scientific, regulatory and economic frameworks, built by many hands working together.
RExPO25 also reinforced the role of REPO4EU as a central platform for this dialogue, and set the stage for the next steps towards making repurposed, precision therapies accessible to patients across Europe.
About RExPO25
RExPO25, the 4th edition of our international series of conferences dedicated to Systems Medicine, Artificial Intelligence and Drug Repurposing, took place in Barcelona on 24-26 September 2025.
Revisit the conference
Curious to learn more? Watch our video compilation for RExPO25