Core insights – Pre-clinical validation of scientific questions
Welcome to REPO4EU: Core insights
Our brand new article series where we bring you closer to our project's core innovations and results.
Preclinical validation
A brief introduction
In the pharmaceutical world, clinical trials play the role of the ultimate “reality check”. However, they are incredibly expensive, time-consuming and involve the most precious variable of all: human lives. Thus, de-risking the path to clinical phase via effective pre-clinical validation lies at the heart of REPO4EU’s ambition.
Within the REPO4EU project, we aim to secure a massive head start for all repurposing projects by providing robust validation of the preclinical hypotheses, generated through advanced bioinformatics, in silico-based target identification, and computational drug predictions. Preclinical validation moves a prediction into living systems, and this exact bridge between computational approaches and mechanistically relevant cellular and animal models, which mimic human disease endotypes, ensures a strong scientific foundation for each drug candidate.
Our goal is to identify the most promising drug repurposing opportunities and ensure that only rigorously validated candidates advance to clinical stages. This approach builds a more precise, safer, and faster highway, accelerating the path from computational discovery to real-world therapeutic impact, while also prioritizing patient safety.
The road so far
From digital blueprints to validated biological evidence
REPO4EU envisions a future where drug repurposing evolves into a precise, mechanism-driven discipline, firmly based on validated biological evidence. The progress achieved so far through database development, validation pilots, and methodological guidelines, directly supports this vision.
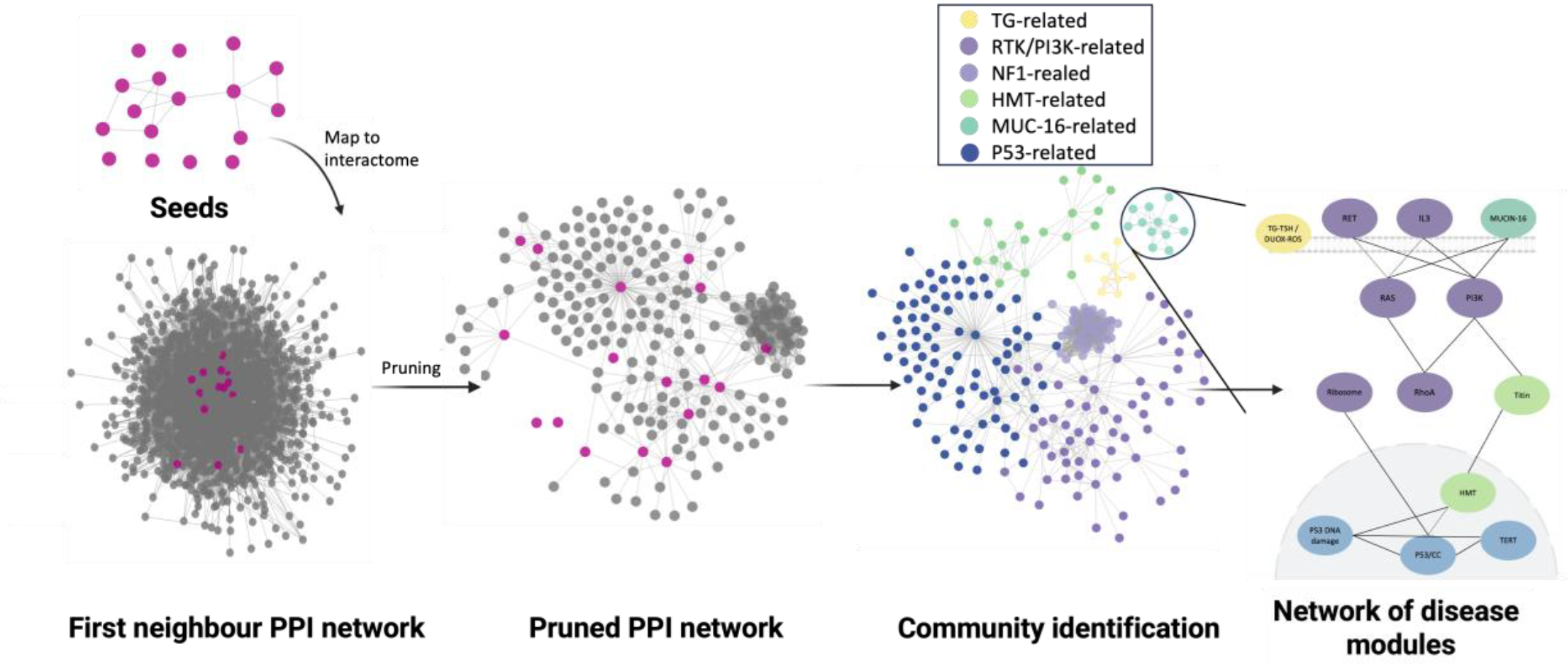
One of our most significant milestones to date is the development of a comprehensive open-access, user-friendly database of cellular and animal disease models, specifically designed to capture the ones with the highest translational potential. This resource acts as a “compass” for researchers, helping them to pinpoint the most appropriate models for validating computational predictions. By strengthening the bridge between in silico predictions and experimental testing, we ensure that the transition is both seamless and scientifically sound.
To demonstrate the power of our approach, REPO4EU has launched validation pilots in three high-stakes medical areas: thyroid cancer, diffuse pontine glioma (DIPG), and infectious diseases. These pilots serve as an essential testing phase providing clinically relevant proof-of-concept studies on repurposing hypotheses before moving to the clinical trials.
Beyond data and testing, we have also developed methodological guidelines for identifying the most appropriate disease models (both cellular and animal), selecting promising repurposable drug candidates, and defining the rationale for combination therapy strategies using a network‑pharmacology approach. Fostering a culture of collaboration, transparency and FAIR (Findable, Accessible, Interoperable and Reusable) data that accelerates the translation of preclinical findings into life-saving clinical scenarios, the guidelines will be made openly accessible to the global scientific community.
Looking ahead
Towards a robust preclinical validation framework
REPO4EU’s commitment to a transformed medical landscape remains the primary force guiding our work. By systematically connecting bioinformatic predictions with clinically relevant models, we are laying the groundwork for an open-access and scientifically rigorous pipeline. Within our platform, the flagship output of our project, stakeholders will be able to access validated tools, disease models, and decision‑making frameworks that bridge the critical gap between computational discovery and clinical application.
Through this infrastructure, we ensure that repurposing strategies are not only innovative but also precise, reproducible, and ready for regulatory consideration. We are redefining the standards for preclinical validation across Europe and beyond, moving away from fragmented research toward a unified, transparent ecosystem.
And we remain laser–focused on our long-term vision, where drug repurposing accelerates therapeutic development, reduces costs, mitigates risks, and ultimately delivers patient-centered solutions to some of the most pressing health challenges worldwide.
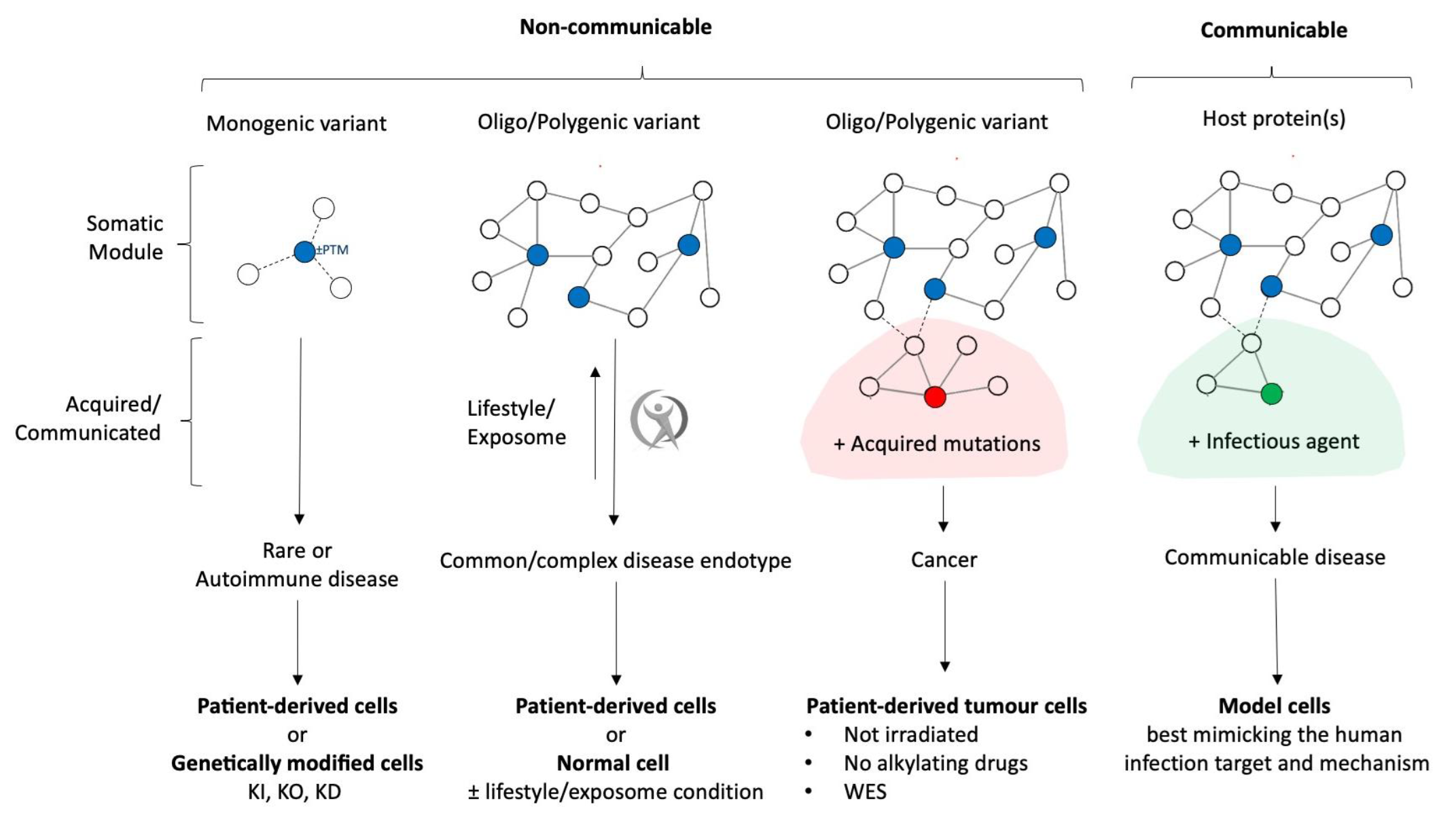
Building on the significant progress achieved so far, we embrace the work required to maximize translational impact as an opportunity for further growth and innovation. Maintaining the translational relevance of disease models and ensuring that cell and animal systems reflect the complexity and diversity of human disease, remains one of our central scientific imperatives. Equally important is the integration of heterogeneous data sources, from computational predictions and preclinical models to clinical datasets, which calls for advanced frameworks for harmonization and interpretation. Another crucial step toward enabling downstream clinical validation is effectively aligning methodologies with regulatory requirements.
Beyond these structural challenges, the scientific landscape itself is constantly evolving. Advances in disease endotyping and in our understanding of drug mechanisms demand ongoing methodological refinement to ensure that preclinical strategies remain up to date and clinically meaningful. Addressing these challenges will be essential to ensure we continue delivering robust, safe, and translatable outcomes that advance the goals of REPO4EU.
Behind the scenes
Meet our multidisciplinary experts’ alliance
The strength of REPO4EU lies in its collaborative engine bringing together diverse expertise to transform computational theories into clinical realities.
Our preclinical validation team, led by Maastricht University, leverages their leading expertise and long-standing experience in preclinical experimentation and animal models to drive reliable validation of drug repurposing candidates. RUMC and UZH contribute specialized knowledge in thyroid cancer and DIPG, providing both animal models and clinical insights that support scientific translation. UHAM contributes essential expertise in viral infectious disease models, while BioLizard ensures methodological statistical rigor to support reliable results. To bridge preclinical data with clinical application, HMPC and 3DPX play a key role in supporting the clinical translation of findings. Finally, GeneSurge is responsible for integrating complex datasets and guiding scientific decision-making.
Collectively, we form a multidisciplinary framework that ensures evidence-based progression of drug candidates.
About our platform
Discover our platform and explore its key features
Become an early tester now!
Follow the link to sign up for our platform's alpha release

